2 melting peaks in real-time PCR - (Jul/16/2006 )
Dear all,
I've had this problem of 2 melt peaks for 2 runs for standard dilution curves in triplicate. Each time the same. The lower concentration dilutions have the lower melt peak (~77) My product is supposed to be ~83.
There's no problem for higher concentration dilutions. What's the problem? Do you think I should reduce primer concnetration or do 5X dilution curves instead of 10X? After gel run there were quite obvious primer dimer bands which seem to get brighter as the dilution curve progresses.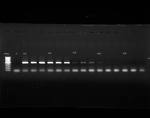
I use SYBR Green I Master mix from Qiagen, Rotor Gene 3000, cDNA samples (2.2ul), primer conc. of 10pmols (0.5ul each) in a 25ul reaction. I have vortexed and spinned down all my reagents well. My -C also shows the melt peak at 77C but my NTCs have no melt peak.
Please help me solve this problem. I don't want to waste more master mixes before I know what's wrong...
Thanks....
Chris.
I can see two possible solutions
1. titrate your primers down...less concentration...see how low you can get them and still get good ampification efficiencies
2. design new primers that don't have quite such an affinity for each other ![]()
what a PIA, good luck
