Useful Dilution Techniques & Calculations - Archived thread (Mar/21/2009 )
I have a pile of bookmarked calculations tools, although I think it's VERY important to learn how to do these so that you understand the principle and are therefore less likely to make errors. I also have a pile of other useful sites in my 'toolbox' bookmarks, here:
https://www.promega.com/biomath/default.htm
https://www.cellbiol.com/scripts/complement...t_sequence.html
https://www.biocarta.com/genes/index.asp
https://www.hprd.org/
https://www.graphpad.com/quickcalcs/Molarityform.cfm
https://www.fauvet.fau.edu/oacm/VetData/Handouts/mouseHO.htm
https://www.sigmaaldrich.com/life-science/c...al-buffers.html
https://info.med.yale.edu/genetics/ward/tavi/PCR.html
https://www.thelabrat.com/protocols/reagents.shtml
https://molbiol.ru/eng/scripts/01_05.html
and, holy moly, whatever you do, don't forget that there's a lot of really good information to be found here: www.protocol-online.org in the main site, not just the forum ![]()
aimikins on Nov 20 2009, 04:49 PM said:
https://www.promega.com/biomath/default.htm
https://www.cellbiol.com/scripts/complement...t_sequence.html
https://www.biocarta.com/genes/index.asp
https://www.hprd.org/
https://www.graphpad.com/quickcalcs/Molarityform.cfm
https://www.fauvet.fau.edu/oacm/VetData/Handouts/mouseHO.htm
https://www.sigmaaldrich.com/life-science/c...al-buffers.html
https://info.med.yale.edu/genetics/ward/tavi/PCR.html
https://www.thelabrat.com/protocols/reagents.shtml
https://molbiol.ru/eng/scripts/01_05.html
and, holy moly, whatever you do, don't forget that there's a lot of really good information to be found here: www.protocol-online.org in the main site, not just the forum
Your site suggestions are quite helpul, but it appears that you are missing out https://nigerianbioscientist.com- It contains a properly categorized listings of free online bioresources- Online tutorials, science databases, online libraries, and lots more. You can check it out right away
bioforum on Mar 21 2009, 07:33 AM said:
So let's start out with definitions:
Dilution: is the mixing of a small accurately measured sample with a large volume of sterile water or normal saline called (diluents or dilution blank)
Laws:
Dilution = V of Sample / Total V of (sample + diluent)
Dilution Factor = Total V of (sample + diluent) / V of sample
** or we can simply say the reciprocal of Dilution
Hi, Does anybody use lipopeptide antigens in immunological reactions? What tests are best suited for them? Can we use MQ water to reconstitute synthetically prepared lipopetides? What about culture medium?
Starting off with this simple example to understand how these laws are applied.

It has been known that if we use a larger volume we obtain a more accurate dilution.
So for better results, we use 1:1000 dilution. And that is by adding 1ml of sample to 999 ml of diluent. But practically we cannot use 999 ml of diluent. So we do what is called a serial dilution.
Serial Dilution: is a dilution made of a series of smaller dilution, and the total dilution is the product of each dilution in the series.
To understand this more, let's see this example.
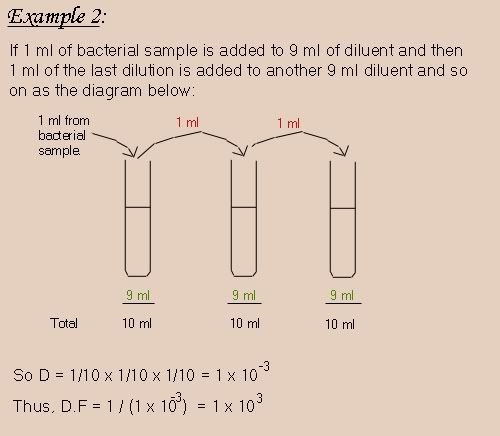

Here's an example combining all of the above.

I hope this brief explanation proves helpful to you...let me know if i've made any mistakes...
<p>thank u for your explanation..
what makes me always confused is the last volume used for inoculation (0.1ml in your example)..</p> <div>-strawberry-</div><hr>
<p>Yeah that's why i almost jumped with 'HAVE' there :blush: sorry about that
i hope my mini-tutorial and examples helped you through it strawberry...
<p>I'm very thankful to you to provide the easier explaination...it's true that many undergrad students have problem in dilution..and the worst thing is if the lecturers/tutors assume that the students are familiar with the concept. it is true that they have learned it in high school, but most of the time they will forget..
Regarding the example 2 given, you dilute a sample (10 ml the final volume) 10x10x10=10 to the power of 3. and u inoculate 0.1 ml in your medium.so supposed the final volume of your medium is 1ml right?
</p> <div>-kent19-</div>
i'm now trying to find how to determine the standard concentration (dially disulphide, diallyl sulphide, ajoene) for Gas chromatography analysis.
and.. sumbody told me to find it, u must calculate from Rf factor..
then i go..." what da heck is Rf? how to calculate it??" ![]()
i try to find from the internet.. but.. nothin.. ![]()
my fren said.. rf is sumthing u must calculate from the graph... then i go..."wut? ![]() graph?
graph? ![]() ?"
?"
GC... for chemistry student... not me.. i havent learn it.. i've only use it twice.. ![]()
please guys.. help me..... how? how? how?
Hello!
I have done serial dilutions of my Ab for IHC, but the staining doesn't make sense. Some lower concentrations have stronger signal the the higher ones. Can you check if I made the right dilutions?
I started with a 1:1000 solution and I need final volumes of 320 uL to each dilution.
For a 1:2000, I took 160 uL from the 1:1000 and add 160 uL of diluting solution.
For a 1:2500, I took 128 uL from the 1:1000 and 192 uL of diluting solution.
And for a 1:3000, I took 107 uL from 1:1000 and 213 uL of diluting sol.
My calculus are wrong?
Thank you very much for your help
Hi all,
I'm new to this website and I found it helpful in reading some of the interested topics
just to mention that I started my undergrad research in purifying RNA mainly H69,
I really need some help in some of the calculations concerning O.D readings
So basically I ran a 1 mL reaction for 4-5 hours, centrifuge to remove any salts, do ethanol precipitation and disolve the pellet in dH2O.
then I take the O.D of the sample and I read it as let say it's 100 O.D at 260nm witha volume of 70 uL
now if I want to make the reading to be 20 OD (since i need to run it in a small 9 by 7 gel) how much of the 100 O.D sample should be taking and how much should i dilute it to.
If there is a website that you think would be helpfull to me to look at, please post it
I will be posting more question, cz I really need help with this as an undergrad
Thanks
Golaso
If you need 20 OD, you simple need to dilute the 100 OD 5 times...
Its best to make a new post with questions like this.
Wish i found this website 1 year ago when i first started in the lab. However i've found it now and so far the content is amazing!
Hello Everyone!
I am kinda confused with the dilutions.....
1.I need to make a 0.02% of Bromophenol blue (working solution) in a 5Xor 10X buffer. I made 5X buffer and then made a 2% firstly.
by adding 0.16 gm to 6.4ml 5X buffer (made 8ml).
Now I need to make a 0.02 or0.01 final dilution. How do i do this?
2. I know the final concentration of my RNA extracted. I need to know how much I am loading or using /ul. I get so annoyed calculating in terms of ug/ul.
Can someone help me please.
Thanks
c1v1=c2v2
concentration, in your case, is weight per volume. g/l=mg/ml=ug/ul. if you know the concentration then you know how much you are loading/ul.
