I'd like your advice on NIH 3T3 sonication - (Feb/07/2012 )
Good day, (please excuse my english, it's not my first language)
I recently started my master less than a year ago and I'm currently trying to do ChIPs on NIH 3T3 cells. My lab's old protocoI requires 25 minutes of sonication and doesn't give really nice chromatin fragments. I decided to try different conditions and I was able to get very nice chromatin fragments (I think so) by sonicating 50e6 cells in 1 ml of 1% SDS nuclei lysis buffer in the diagenode bioruptor (2*5 cycles of 30s on/30s off). Here's the timecourse I did.
However, I would need to dilute 1:10 my chromatin to IP in 0.1% SDS. That would mean 3.2 ml for 16e6 cells, wich is way too much. I'd like advices from people who usually do ChIP on NIH 3T3. What is your sonication buffer? How long do you have to sonicate? How many cell do you sonicate at a time and in what volume? Feedback would be greatly appreciated.
Thank you very much!
Joël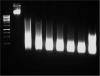
About the image:
2nd track is non sonicated chromatin,
3rd is 5 minutes
4th is 10 minutes and so on...
The last track is non purified sonicated chromatin put directly on the gel.
Have you tried lower concentrations of SDS. Looking at your sheering profiles on the gel it looks like you have some room to optimize. Also you might find that your pulldown is improved with lower SDS concentrations if you're looking for epitopes other than histones or histone mods.
