miRNA northern - (Jan/26/2010 )
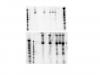
im trying to run a northern on miRNA and since U6 and 5S rRNA seems to be the most widely used endogenous control,
I have made probe with 32P and ran northern on 12.5% Urea-polyacrylamide gel. Everything went fine, i think,
and I got a clear band at around 95 nucleotide (based on the distance i compared on Decade Marker-Ambion).
However, the problem is that U6 is suppose to be, as far as i know, at around 115 nt in length.
Additionally, when i stripped the same membrane and re-probed using 5s rRNA, i get the band of correct size for
5s rRNA, at around 115 nucleotide, but the previous U6 band seems to have gotten thicker.
I have left the membrane in -20'C in casette to overcome the drying issue, and have used
2x SSC, 0.1% SDS solution in 70'C for stripping for 1 hour.
Any suggestions on what i'm doing wrong ????
first picture is the U6, and 5s on the bottom after stripping and re-probing.

leaving in cassette is not a great idea but leave it there in a sealed bag. so second time around due to nonspecific binding it might have incrased or the exposure time/intensifying screen use/using a better film also changes it...we have seen kodak films are atleast 10 times sensitive than the rest cheaper ones available...
I strip my blots in the hybridization tube at 80C in the oven twice for 15 min each and they are clean completely...same buffer as you have mentioned here...if you work with plant mirna I have got the same size for U6 but I donot know what the real size is. One expert told me in person it is around 100nt. I got around 90-95nt size too - with palnt specific U6 probe...
